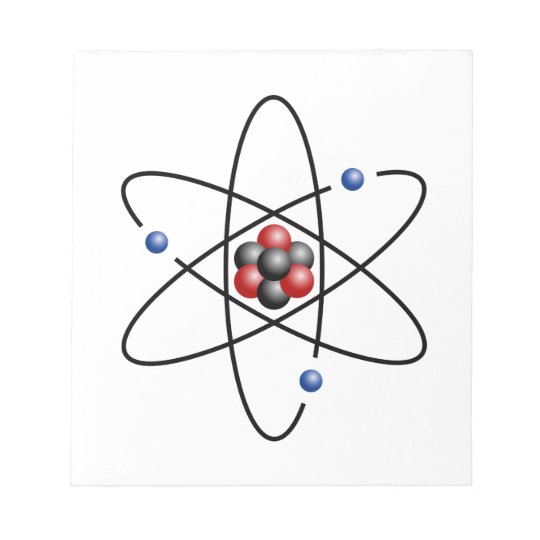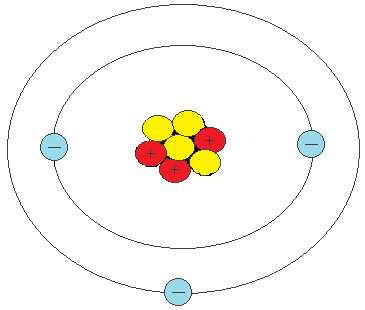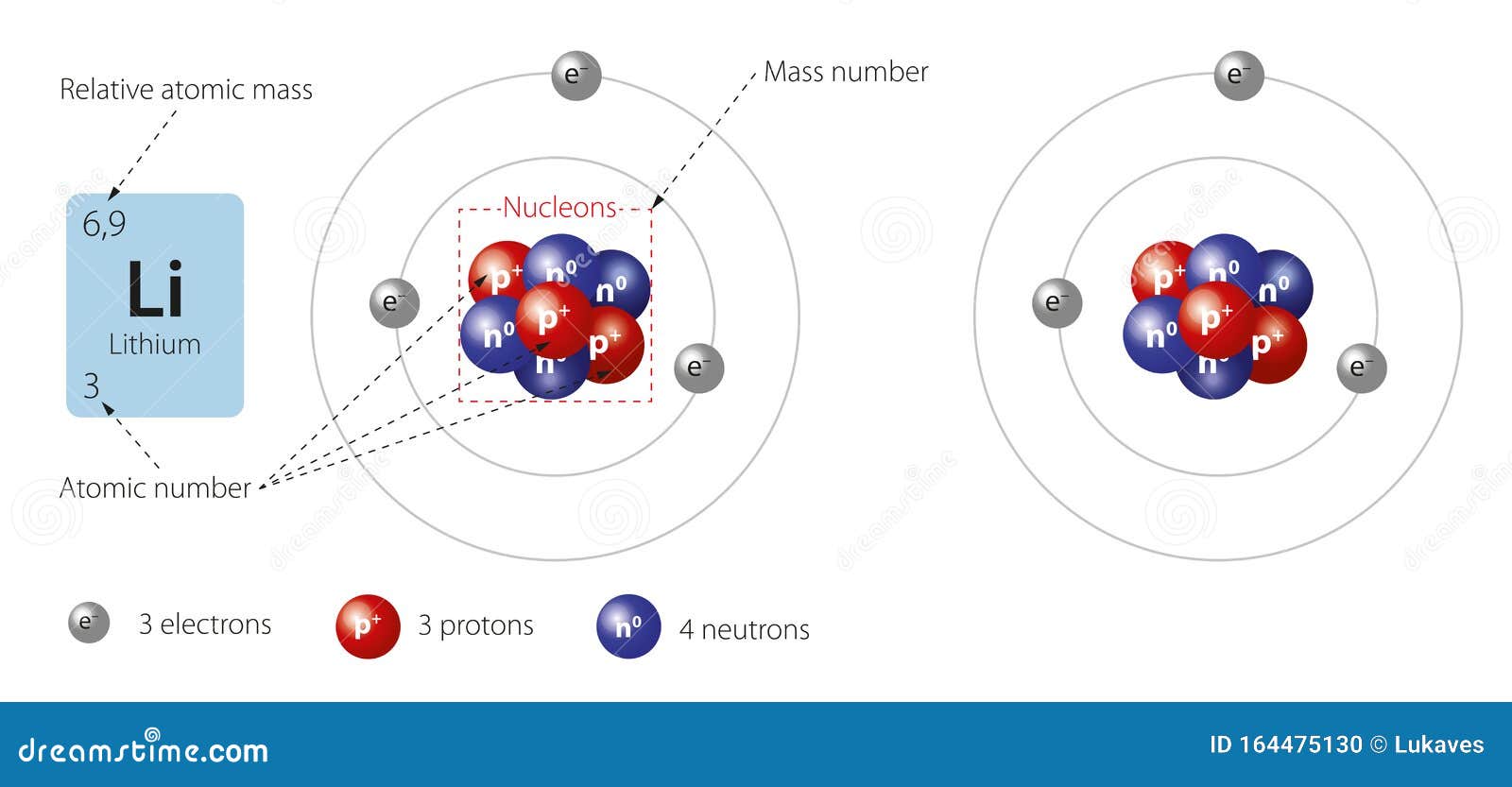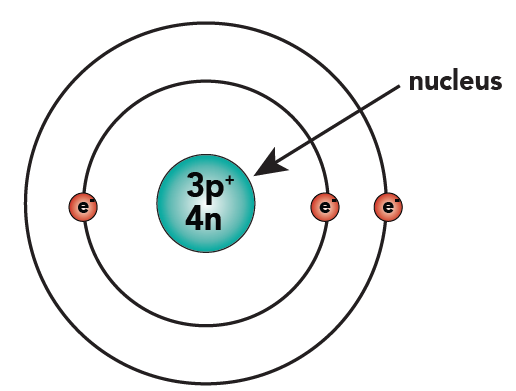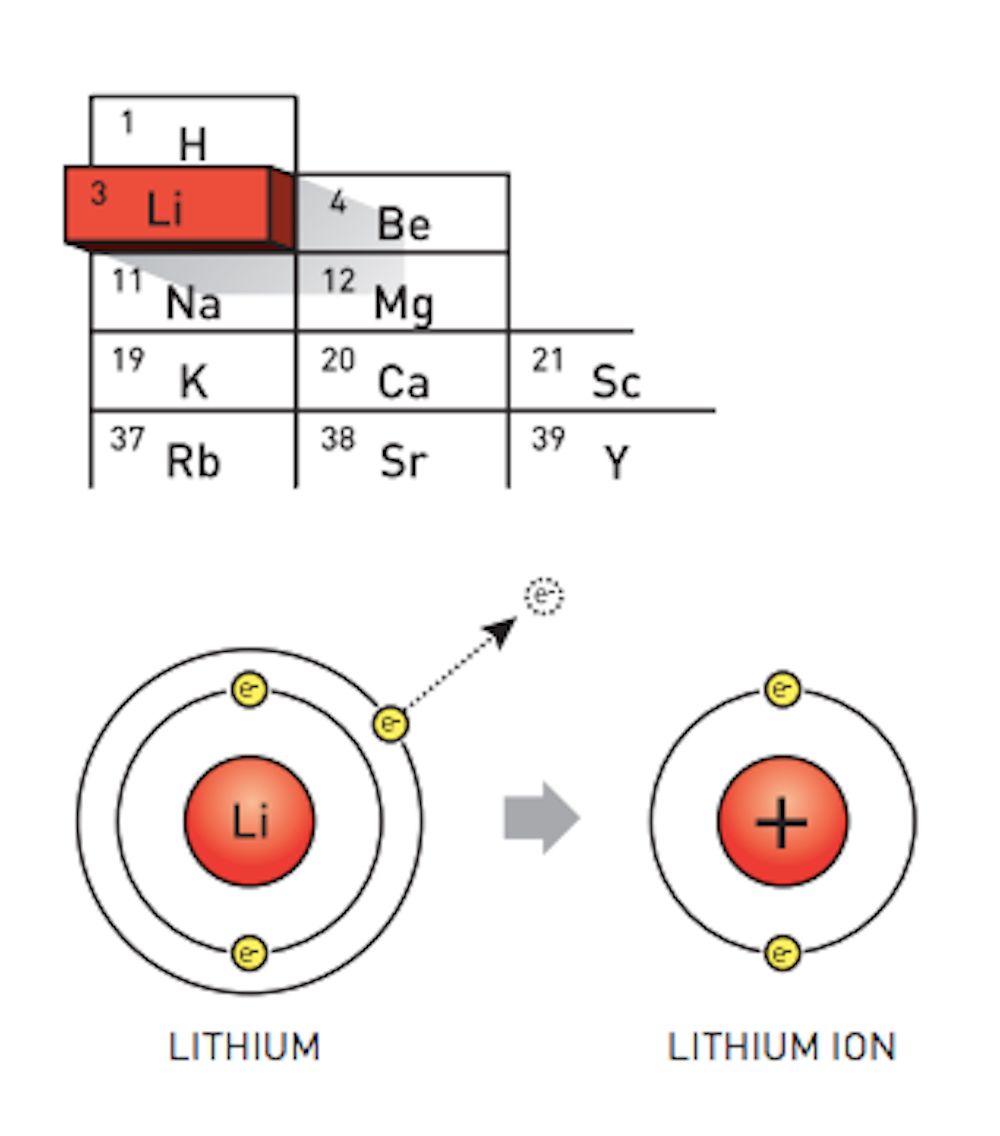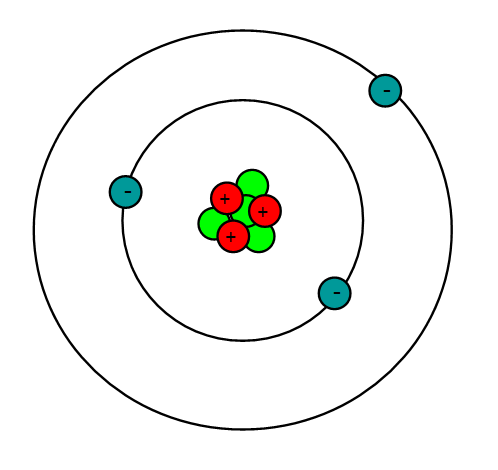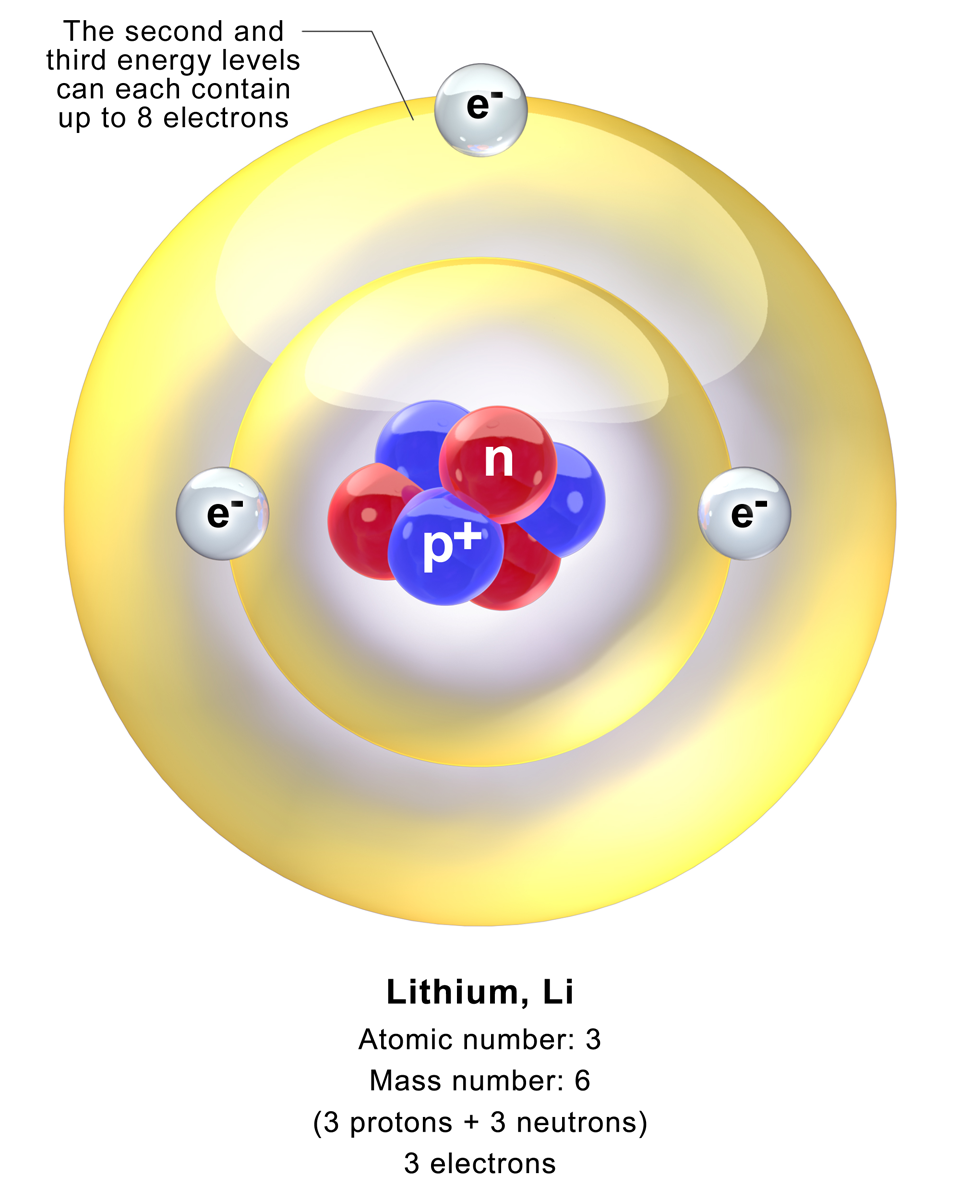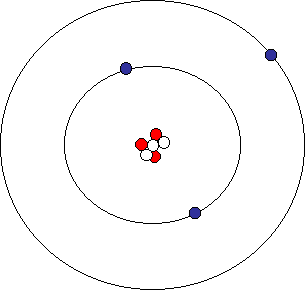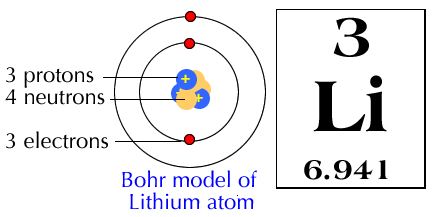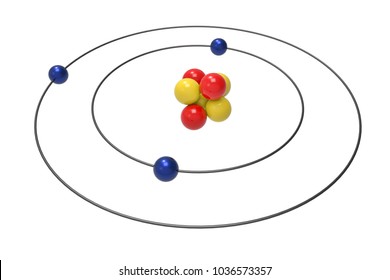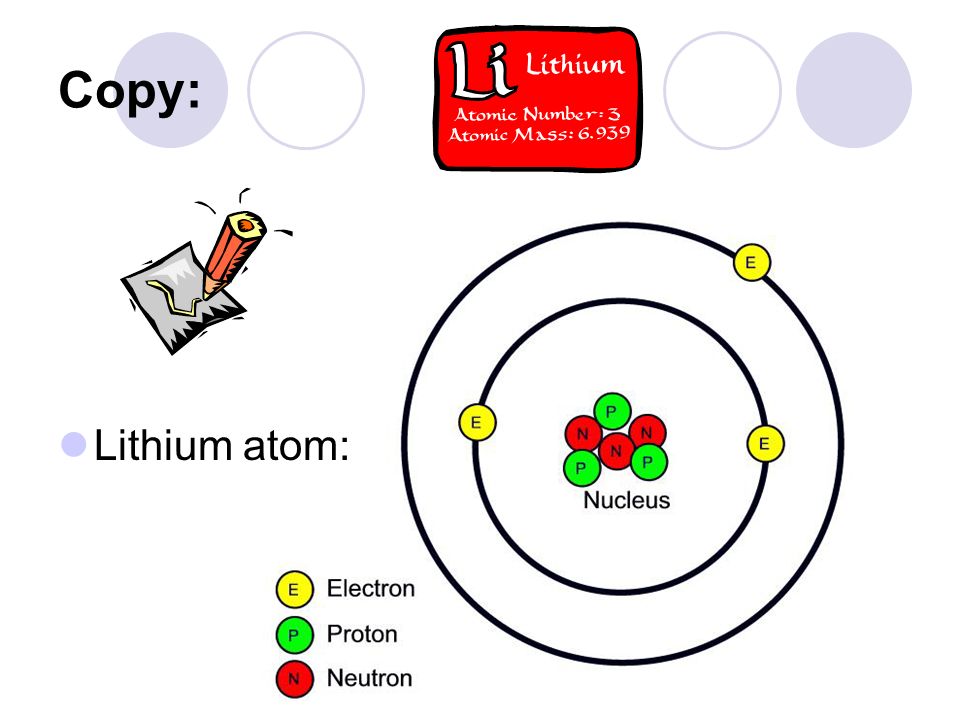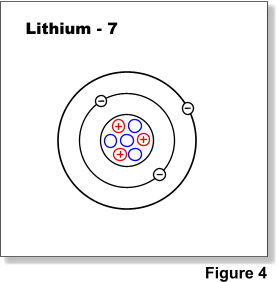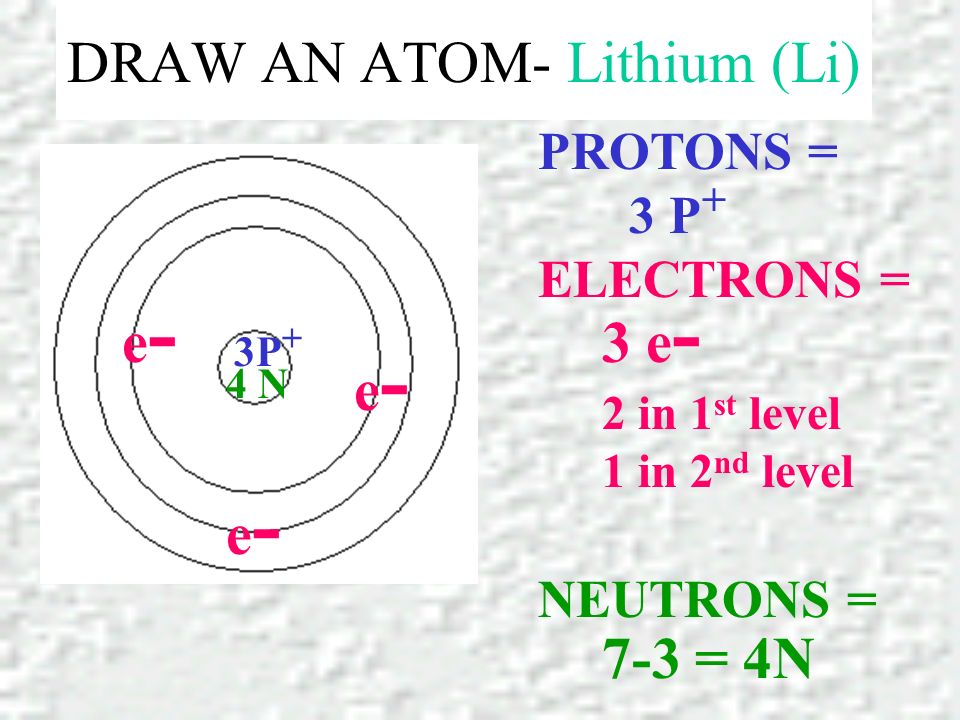
PRACTICE DRAWING ATOMS. DRAWING ATOMS RULES PROTONS = Atomic number ELECTRONS = Atomic number NEUTRONS = mass number – atomic number 1 st level can hold. - ppt download

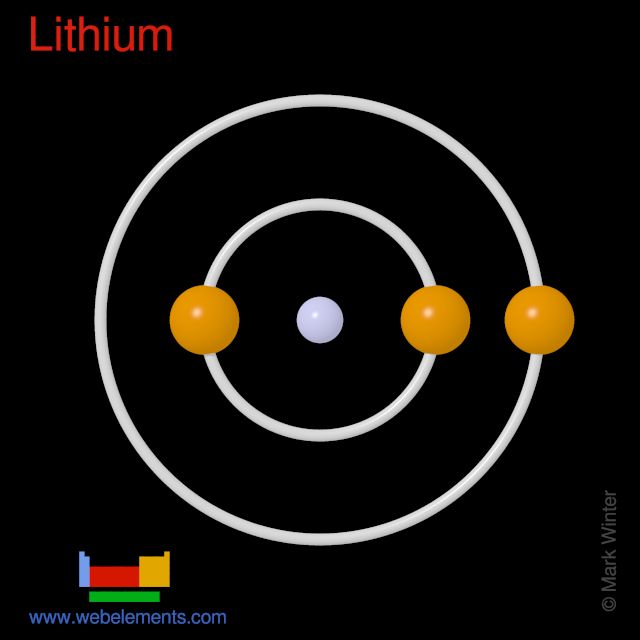
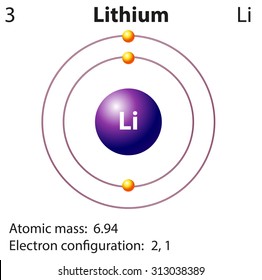
Li Lithium Element Information: Facts, Properties, Trends, Uses and comparison - Periodic Table of the Elements | SchoolMyKids